Cost-Effectiveness and Optimal Control of Hantavirus Transmission in Rodent Populations
Main Article Content
Abstract
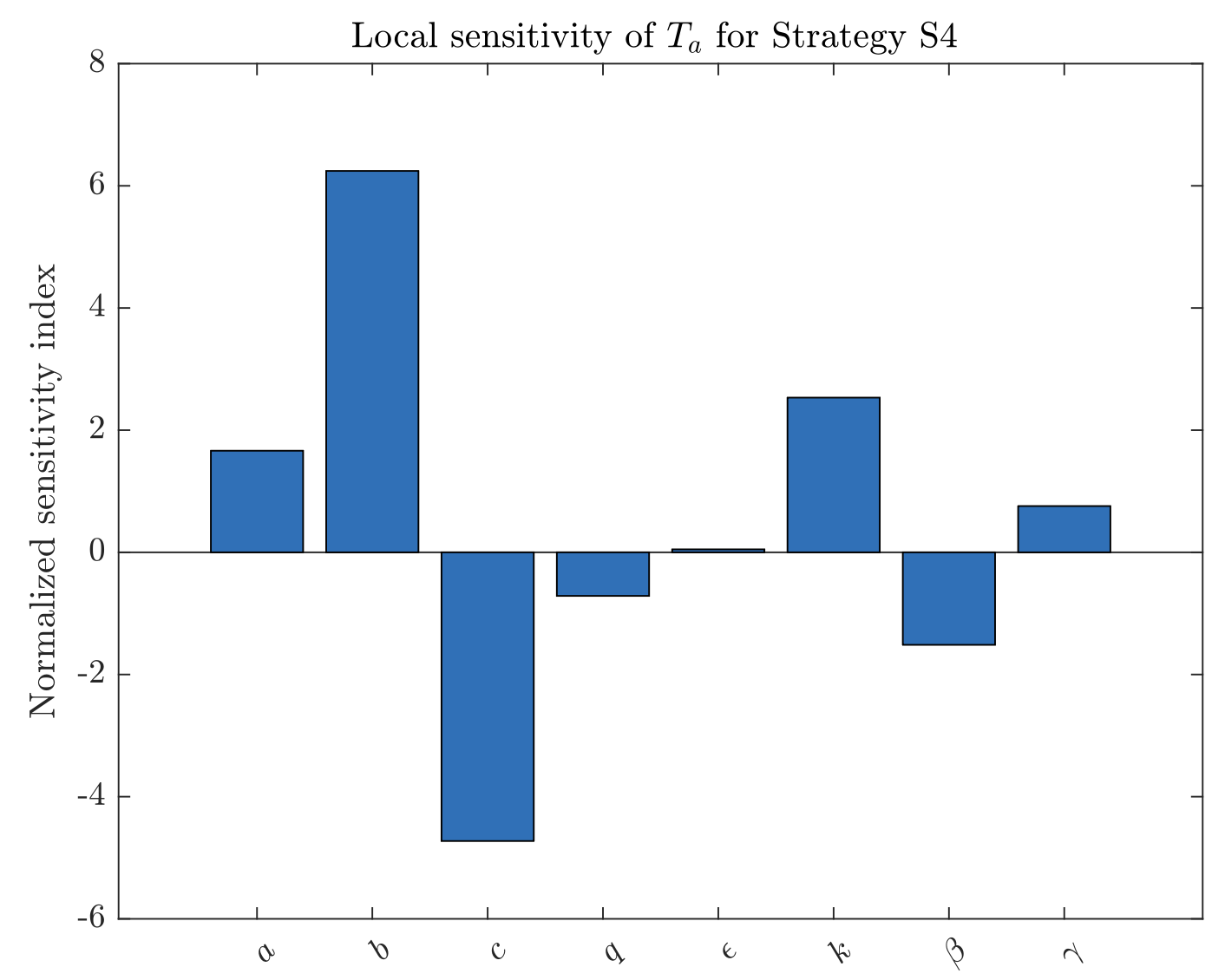
In this paper, we formulate and analyze a deterministic optimal control model for the transmission dynamics of hantavirus infection in rodent populations and identify economically efficient intervention strategies. The model incorporates three time-dependent controls: rodent harvesting, transmission reduction, and alien-oriented control. By applying Pontryagin’s Maximum Principle, we derive the Hamiltonian, the adjoint system, and explicit characterizations of the optimal controls, leading to the corresponding optimality system. Numerical solutions are obtained using a forward-backward sweep algorithm. Simulation results demonstrate that combined interventions can substantially reduce the infected rodent population; however, under limited resources, selecting a cost-effective policy is crucial. A health-economic evaluation based on the average and incremental cost-effectiveness ratios (ACER and ICER) shows that the joint implementation of harvesting and transmission reduction provides the most effective strategy among the alternatives considered. We further present a local normalized sensitivity analysis, highlighting the parameters that most strongly influence the predicted infections averted. These findings support the adoption of integrated, rodent-focused interventions for mitigating hantavirus transmission and offer quantitative guidance for informed public health decision-making.
Article Details

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
References
[1] Li Y, Xiao Y. Effects of nonlinear impulsive controls and seasonality on hantavirus infection. Mathematical Biosciences. 2025 feb;380:109378. doi:10.1016/j.mbs.2025.109378.
[2] Tessema Alemneh H, et al Makinde. Mathematical Modelling of MSV Pathogen Interaction with Pest Invasion on Maize Plant. Global Journal of Pure and Applied Mathematics. 2019;15(1):55-79.
[3] Peter OJ, Panigoro HS, Ibrahim MA, Otunuga OM, Ayoola TA, Oladapo AO. Analysis and dynamics of measles with control strategies: a mathematical modeling approach. International Journal of Dynamics and Control. 2023 oct;11(5):2538-52. doi:10.1007/s40435-022-01105-1.
[4] Berhe HW. Optimal Control Strategies and Cost-effectiveness Analysis Applied to Real Data of Cholera Outbreak in Ethiopia’s Oromia Region. Chaos, Solitons and Fractals. 2020 sep;138:109933. doi:10.1016/j.chaos.2020.109933.
[5] Alqahtani Z, Almuneef A, El-Shahed M. Mathematical Model for the Control of Red Palm Weevil. Axioms. 2024 sep;13(9):637. doi:10.3390/axioms13090637.
[6] Alnafisah Y, El-Shahed M. Optimal control of red palm weevil model incorporating sterile insect technique, mechanical injection, and pheromone traps. Alexandria Engineering Journal. 2024;93:382-91. doi:10.1016/j.aej.2024.02.059.
[7] El-Mesady A, Elsadany AA, Mahdy AMS, Elsonbaty A. Nonlinear dynamics and optimal control strategies of a novel fractional-order lumpy skin disease model. Journal of Computational Science. 2024;79:102286. doi:10.1016/j.jocs.2024.102286.
[8] Oguntolu FA, Peter OJ, Omede BI, Balogun GB, Ajiboye AO, Panigoro HS. Mathematical Modeling on the Transmission Dynamics of Diphtheria with Optimal Control Strategies. Jambura Journal of Biomathematics (JJBM). 2025;6(1):1-22. doi:10.37905/jjbm.v6i1.29716.
[9] Wanjala HM, Okongo MO, Ochwach JO. Mathematical Model of the Impact of Home-Based Care on Conta- gious Respiratory Illness Under Optimal Conditions. Jambura Journal of Biomathematics (JJBM). 2024;5(2):83-94. doi:10.37905/jjbm.v5i2.27611.
[10] Ludji DG, Hurit RU, Manek SS, Ndii MZ. Comparison of Optimal Control Effect from Fungicides and Pseu- domonas Fluorescens on Downy Mildew in Corn. Jambura Journal of Biomathematics (JJBM). 2024;5(1):38-45. doi:10.37905/jjbm.v5i1.23153.
[11] Al Ajlan ZS, El-Shahed M. Mathematical Modeling of the Impact of Desert Dust on Asthma Dynamics. Axioms. 2025;14(8):639. doi:10.3390/axioms14080639.
[12] Afolabi AS, Miswanto M. Mathematical Modeling, Optimal Control and Cost-Effectiveness Analysis of Diphtheria Trans- mission Dynamics. Jambura Journal of Biomathematics (JJBM). 2025;6(2):88-108. doi:10.37905/jjbm.v6i2.30851.
[13] Jose SA, Raja R, Alzabut J, Rajchakit G, Cao J, Balas VE. Mathematical modeling on transmission and optimal control strategies of corruption dynamics. Nonlinear Dynamics. 2022;109(4):3169-87. doi:10.1007/s11071-022-07581-6.
[14] Alnafisah Y, El-Shahed M. Stochastic Analysis of a Hantavirus Infection Model. Mathematics. 2022;10(20):3756. doi:10.3390/math10203756.
[15] Kocabiyik M, Ongun MY. Distributed order hantavirus model and its nonstandard discretizations and stability analysis. Mathematical Methods in the Applied Sciences. 2025;48(2):2404-20. doi:10.1002/mma.10442.
[16] Moustafa M, Hafiz Mohd M, Izani Ismail A, Aini Abdullah F. Dynamical Analysis of a Fractional-Order Han- tavirus Infection Model. International Journal of Nonlinear Sciences and Numerical Simulation. 2020;21(2):171-81. doi:10.1515/ijnsns-2018-0292.
[17] Moustafa M, Abdullah FA, Shafie S, Ismail Z. Dynamical behavior of a fractional-order Hantavirus infection model incorporating harvesting. Alexandria Engineering Journal. 2022;61(12):11301-12. doi:10.1016/j.aej.2022.05.004.
[18] Supriatna AK, Napitupulu H, Ndii MZ, Ghosh B, Kon R. A Mathematical Model for Transmission of Hantavirus among Rodents and Its Effect on the Number of Infected Humans. Computational and Mathematical Methods in Medicine. 2023;2023(1):9578283. doi:10.1155/2023/9578283.
[19] Mohamed Yusof F, Abdullah FA, Md Ismail AI. Modeling and Optimal Control on the Spread of Hantavirus Infection. Mathematics. 2019 dec;7(12):1192. doi:10.3390/math7121192.
[20] Yusof FM, Folarin FM. Modeling the transmission dynamics of Hantavirus infection under the effect of vaccination and other optimal controls. Discovering Mathematics. 2023;45(1):56-75.
[21] Mohamed Yusof F, Md Ismail AI, Abu Hassan AY. Implication of Predator Interaction of the Spread of Hantavirus Infection. Matematika. 2018:205-26. doi:10.11113/matematika.v34.n2.953.
[22] Peixoto ID, Abramson G. The effect of biodiversity on the hantavirus epizootic. Ecology. 2006;87(4):873-9. doi:10.1890/0012-9658(2006)87[873:TEOBOT]2.0.CO;2.
[23] Rodrigues P, Silva CJ, Torres DFM. Cost-Effectiveness Analysis of Optimal Control Measures for Tuberculosis. Bulletin of Mathematical Biology. 2014;76(10):2627-45. doi:10.1007/s11538-014-0028-6.
[24] Berhe HW, Gebremeskel AA, Melese ZT, Al-arydah M, Gebremichael AA. Modeling and global stability analysis of COVID-19 dynamics with optimal control and cost-effectiveness analysis. Partial Differential Equations in Applied Mathematics. 2024;11:100843. doi:10.1016/j.padiff.2024.100843.
[25] Lenhart S, Workman JT. Optimal Control Applied to Biological Models. Chapman and Hall/CRC; 2007. doi:10.1201/9781420011418.